Instead, you are to know the bond angles for the basic geometries and understand where a bond angle will be smaller or greater than expected based on the existence of a lone pair or a multiple bond around a central atom. The basic geometry is trigonal planar with 120° bond angles, but we see that the double bond causes slightly larger angles (121°), and the angle between the single bonds is slightly smaller (118°).Īlthough in both formaldehyde (H 2CO) and ammonia (NH 3) examples exact bond angles are given, you are not expected to memorize the bond angles in EVERY molecule. This molecule has regions of high electron density that consist of two single bonds and one double bond. 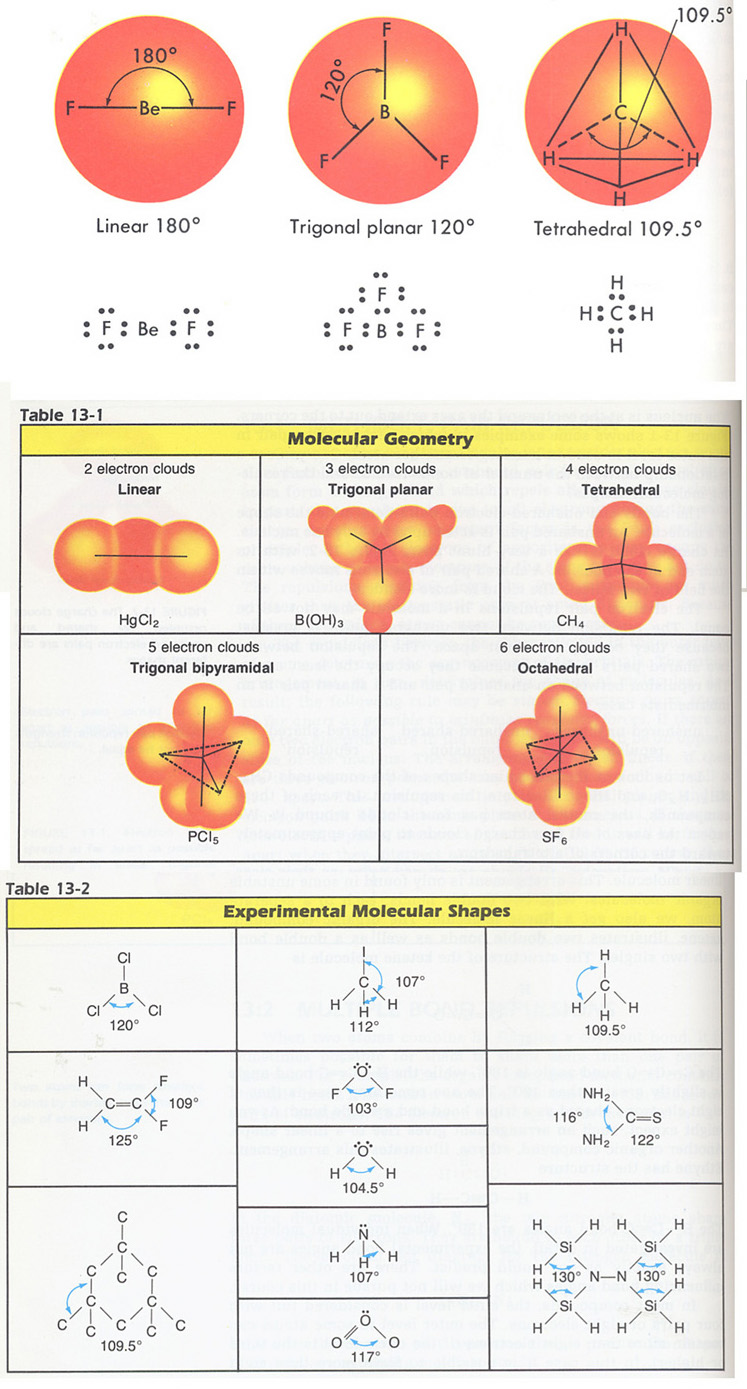
The H–N–H bond angles in NH 3 are slightly smaller than the 109.5° angle in a regular tetrahedron because the lone pair-bonding pair repulsion is greater than the bonding pair-bonding pair repulsion.Ĭonsider formaldehyde, H 2CO, which is used as a preservative for biological and anatomical specimens. Again, there are slight deviations from the ideal because lone pairs occupy larger regions of space than do bonding electrons. The ideal bond angles in a trigonal pyramid are based on the tetrahedral electron pair geometry. In the ammonia molecule, the three hydrogen atoms attached to the central nitrogen are not arranged in a flat, trigonal planar molecular structure, but rather in a three-dimensional trigonal pyramid with the nitrogen atom at the apex and the three hydrogen atoms forming the base. Lone pair > triple bond > double bond > single bond The order of sizes from largest to smallest is: A lone pair of electrons occupies a larger region of space than the electrons in a triple bond in turn, electrons in a triple bond occupy more space than those in a double bond, and so on. This order of repulsions determines the amount of space occupied by different regions of electrons. Lone pair-lone pair > lone pair-bonding pair > bonding pair-bonding pair The order of electron-pair repulsions from greatest to least repulsion is: VSEPR theory predicts these distortions by establishing an order of repulsions and an order of the amount of space occupied by different kinds of electron pairs. (c) The actual bond angles deviate slightly from the idealized angles because the lone pair takes up a larger region of space than do the single bonds, causing the HNH angle to be slightly smaller than 109.5°.Īs seen in Figure 2, small distortions from the ideal angles for a given electron-pair geometry can result from differences in repulsion between various regions of electron density. (b) The trigonal pyramidal molecular structure is determined from the electron-pair geometry. (a) The electron-pair geometry for the ammonia molecule is tetrahedral with one lone pair and three single bonds. VSEPR structures like this one are often drawn using the wedge and dash notation, in which solid lines represent bonds in the plane of the page, solid wedges represent bonds coming up out of the plane, and dashed lines represent bonds going down into the plane. The molecular structure of the methane molecule, CH 4, is shown with a tetrahedral arrangement of the hydrogen atoms. One of these regions, however, is a lone pair, which is not included in the molecular structure, and this lone pair influences the shape of the molecule (Figure 2). On the other hand, the ammonia molecule, NH 3, also has four electron pairs associated with the nitrogen atom, and thus has a tetrahedral electron-pair geometry. 
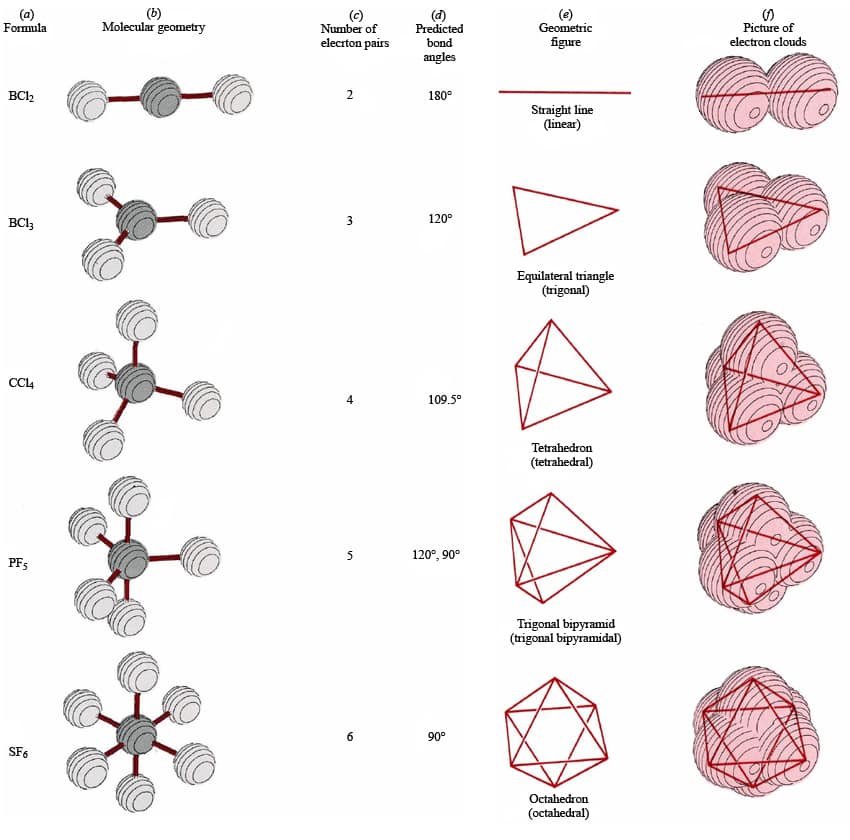
The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom.įor example, the methane molecule, CH 4, which is the major component of natural gas, has four bonding pairs/domains of electrons around the central carbon atom the electron-pair geometry is tetrahedral, as is the molecular structure (Figure 1). The structure that includes only the placement of the atoms in the molecule is called the molecular structure. We differentiate between these two situations by naming the geometry that includes all electron pairs the electron-pair geometry. So all central atoms have one of the defined geometries above (linear, → octahedral) and then if it has a number of lone pairs greater than zero it has a different molecular structure/shape. 
It shows the overall shape that the bonds make within a structure that still abides by a set electron-pair geometry defined above. Molecular structure/shape describes the location of the atoms, not the electrons. The electron-pair geometries shown in the previous page describe all regions where electrons are located, bonds as well as lone pairs. It is important to note that electron-pair geometry around a central atom is not the same thing as its molecular structure/shape. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
